
Home | About Us | JCPS Home | EHS Science Video
- Chemistry Topics: 1) Matter and Measurement, 2) Atoms, Molecules, and Ions, 3) Stoichiometry, 4) Aqueous Solutions, 5) Thermochemistry, 6) Periodic Properties, 7) Solids, Liquids, and Gases, 8) Chemical Bonding, 9) Molecular Geometry, 10) Properties of Solutions, 11) Chemical Kinetics, 12) Chemical Equilibrium, 13) Acid-Base Chemistry, 14) Thermodynamics, 15) Electrochemistry, 16) Nuclear Chemistry
Sodium
By: William Gray

History
Sodium is a chemical element with the symbol Na (from Latin: natrium) in the periodic table and atomic number 11. It is a soft, silvery-white, highly reactive metal and is a member of the alkali metals; its only stable isotope is 23Na. Salt has been an important commodity in human activities, as shown by the English word salary, which derives from salarium, the wafers of salt sometimes given to Roman soldiers along with their other wages. In medieval Europe, a compound of sodium with the Latin name of sodanum was used as a headache remedy. The name sodium is thought to originate from the Arabic suda, meaning headache, as the headache-alleviating properties of sodium carbonate or soda were well known in early times.[27] The chemical abbreviation for sodium was first published by Jöns Jakob Berzelius in his system of atomic symbols,[28] and is a contraction of the element's new Latin name natrium, which refers to the Egyptian natron,[27] a natural mineral salt primarily made of hydrated sodium carbonate. Natron historically had several important industrial and household uses, later eclipsed by other sodium compounds. Although sodium, sometimes called soda, had long been recognised in compounds, the metal itself was not isolated until 1807 by Sir Humphry Davy through the electrolysis of sodium hydroxide.[29][30]
Sodium imparts an intense yellow color to flames. As early as 1860, Kirchhoff and Bunsen noted the high sensitivity of a sodium flame test, and stated in Annalen der Physik und Chemie:[31]
Physical properties

Sodium is a soft, silvery-white, highly reactive metal. Solid, the density is 0.968−3 gcm. 207.9 °F. Atomic radius is 186 pm.
Chemical properties
The electronegativity is 0.93 (Pauling scale). Ionization energy is 495.8 kJ·mol−1.

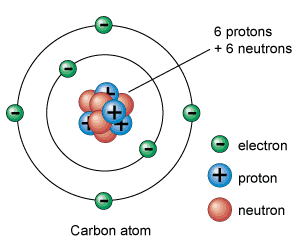
http://www.google.com/imgres?um=1&hl=en&tbo=d&biw=1600&bih=673&tbm=isch&tbnid=vuxUSksqlRA1nM:&imgrefurl=http://www.universetoday.com/56637/atom-model/&docid=O3dtI3kfT11hFM&imgurl=http://ut-images.s3.amazonaws.com/wp-content/uploads/2010/02/c-atom_e1.gif&w=300&h=250&ei=qyibUOmdAoim8ATq7ICQBQ&zoom=1&iact=hc&vpx=1317&vpy=212&dur=93&hovh=200&hovw=240&tx=162&ty=140&sig=106956729340821256264&page=1&tbnh=136&tbnw=163&start=0&ndsp=27&ved=1t:429,r:6,s:20,i:220
http://en.wikipedia.org/wiki/Sodium